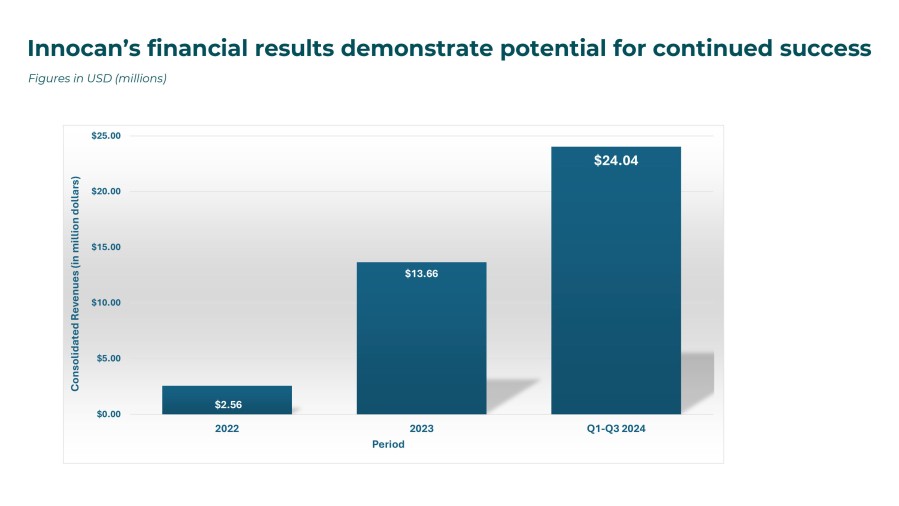
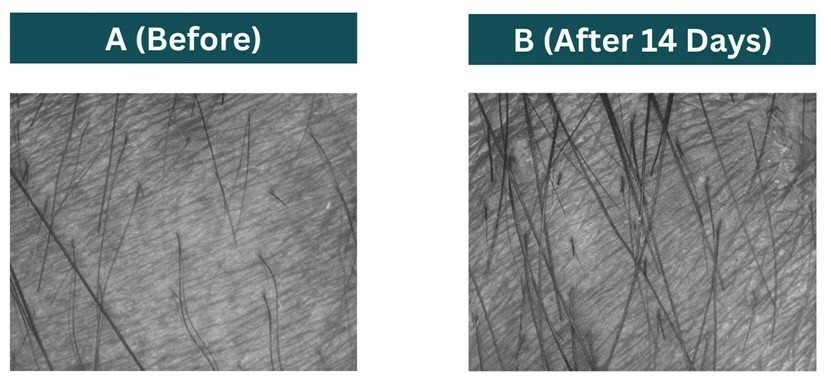
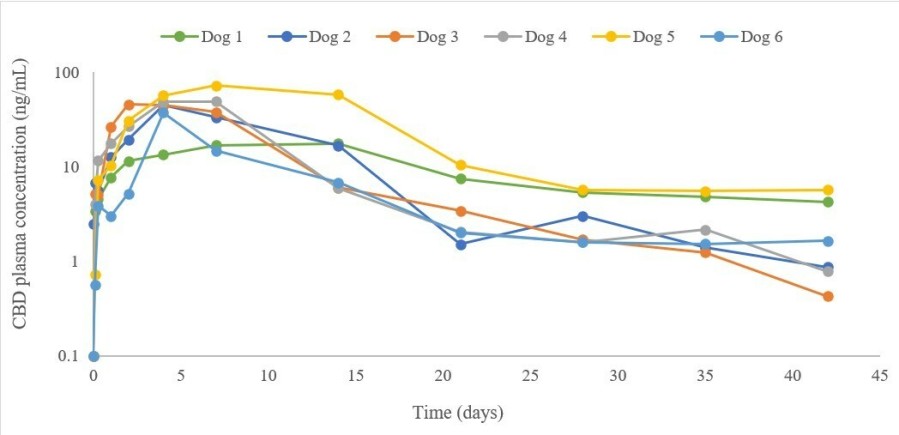
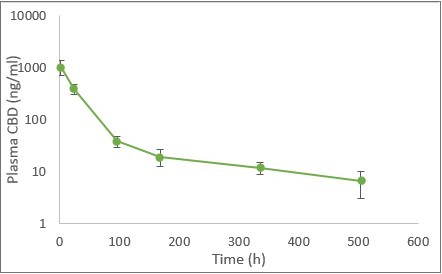
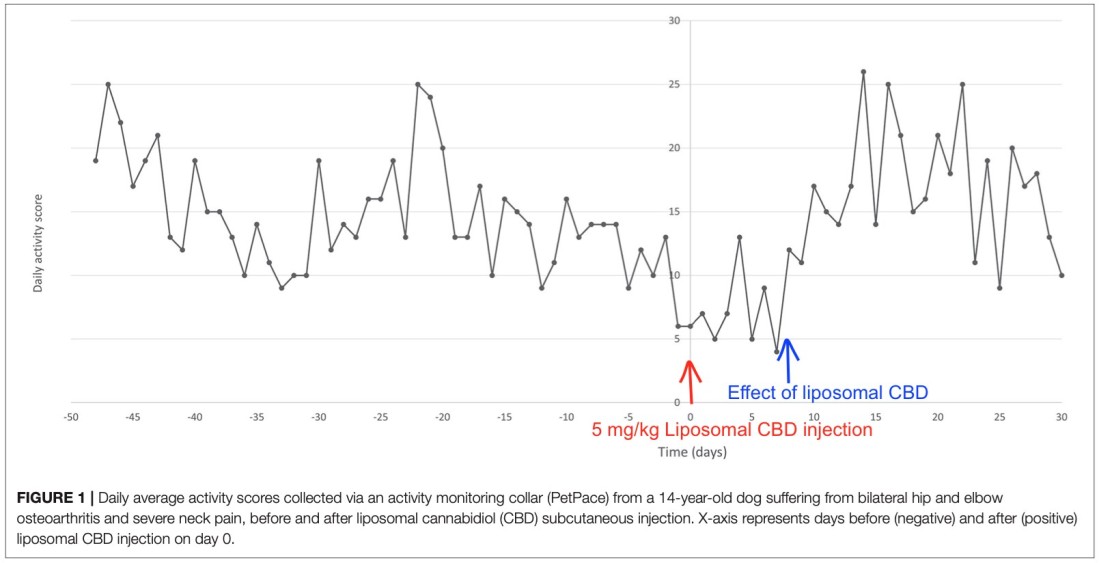
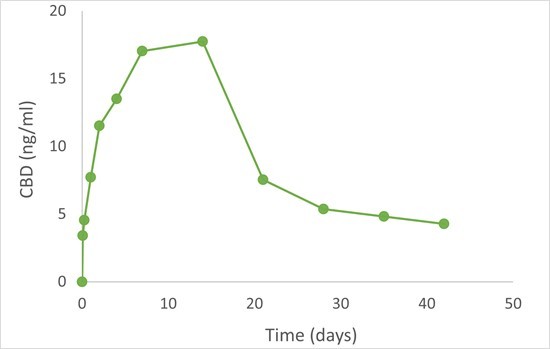
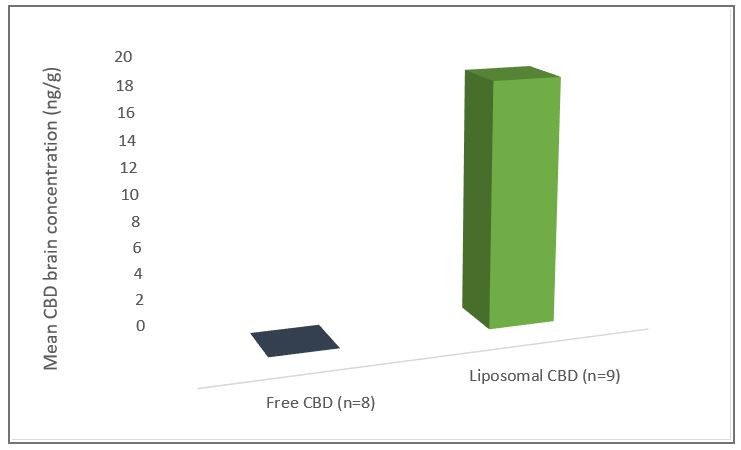
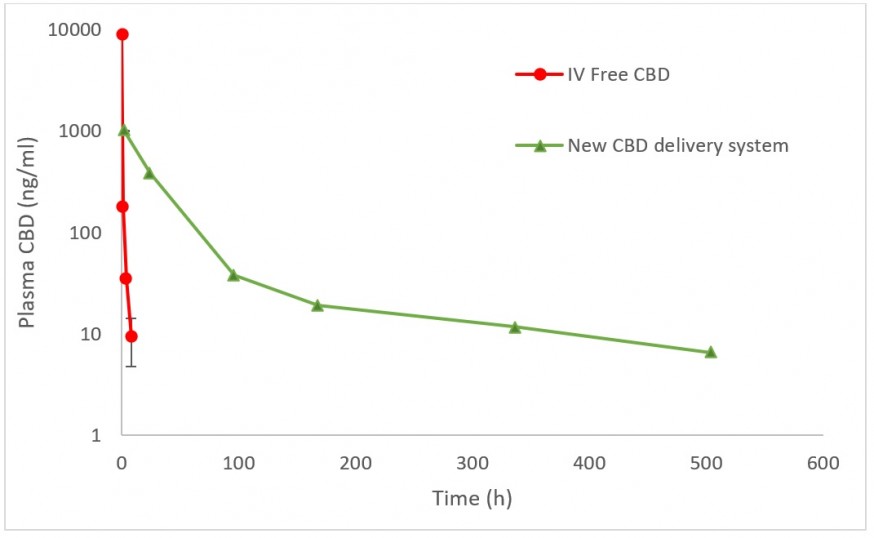
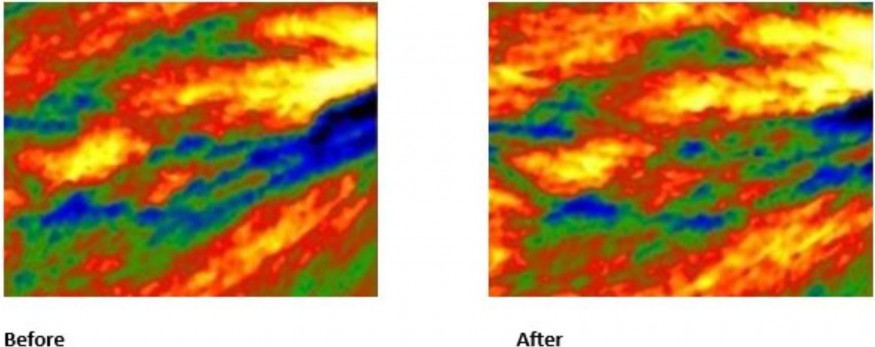
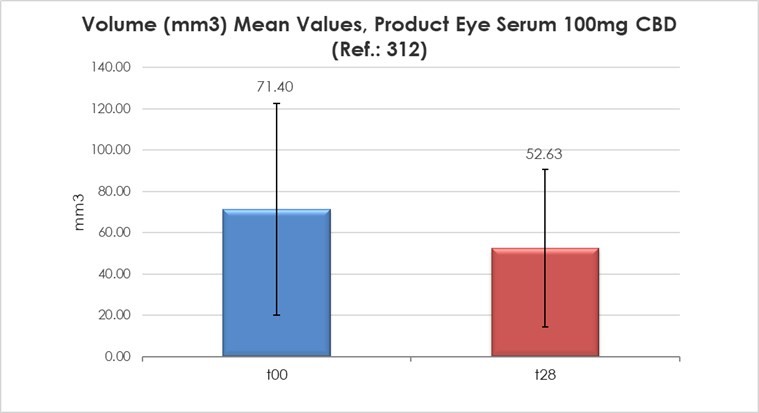
HERZLIYA, Israel and CALGARY, AB , Dec. 10, 2025 /CNW/ -- Innocan Pharma Corporation (" Innocan " or the " Company ") (CSE: INNO) (FSE: IP4) (OTCQB: INNPF) is pleased to announce that, further to its press release of July 23, 2025 , it has publicly filed an amendment to its registration statement on Form F-1 with the U.S. Securities and Exchange Commission (the " SEC ") relating to a proposed public offering of units (each,... Read More